How Long Have Babies Been Getting the Hepatitis B Vaccine
Fundamental information
| Manner of transmission | Contact with infected claret or body fluids during childbirth (vertical transmission); sexual intercourse, intravenous drug employ or contact with broken skin (horizontal transmission). |
|---|---|
| Incubation menses | 45–180 days, ordinarily sixty–90 days. |
| Period of communicability | Potentially infectious ii–3 weeks before the onset of symptoms, during the clinical illness and usually for 2–3 months after astute hepatitis B affliction; or for as long as HBsAg continues to exist present in blood (chronic hepatitis B carrier country). |
| Incidence and burden of disease | New Zealand is a country with a low overall prevalence of hepatitis B railroad vehicle, but it contains sure populations with high prevalence. All pregnant women and loftier-run a risk groups should be screened for chronic HBV infection. HBV conquering in infancy is very likely to atomic number 82 to chronic infection. Chronic HBV infection tin can progress to cirrhosis and liver cancer. |
| Funded vaccines | HepB (Engerix-B) DTaP-IPV-HepB/Hib (Infanrix-hexa). |
| Dose, presentation, road | HepB:
DTaP-IPV-HepB/Hib:
|
| Funded vaccine indications and schedule | At ages 6 weeks, three months and v months: DTaP‑IPV‑HepB/Hib. Infants born to HBsAg-positive mothers should receive HepB vaccine plus HBIG at nascence, and so the usual childhood schedule. Serological testing at age nine months (anti-HBs and HBsAg). Individuals with eligible conditions or close household contacts of infected individuals: HepB (encounter department 9.5). |
| Recommended, unfunded | Those with increased risk from occupational or sexual exposure to body fluids and faeces, or receiving regular blood products. Those with developmental disability, electric current or prior injectable drug users, prison house inmates, and travellers to and from high-prevalence countries. |
| Vaccine effectiveness | In general, efficacy is 85–95 per centum in high chance groups, though probable to be lower in older individuals and those with immunocompromise. Protection is expected to be lifelong and boosters are non required. |
| Public wellness measures | Notify all cases of astute hepatitis B infection (see section 9.8). |
9.1. Virology
The hepatitis B virus (HBV) is a partially double-stranded Deoxyribonucleic acid virus belonging to the Hepadnaviridae family. Three major subunits make up the structural components:
- the HBV genome, a small-scale, circular, partially double-stranded DNA molecule, in association with a polymerase enzyme
- the nucleocapsid cadre, which surrounds the genome and consists of core protein (hepatitis B cadre antigen, HBcAg)
- the outer lipoprotein envelope, which contains the hepatitis B surface antigen (HBsAg).
The genome has four genes (S, C, 10 and P). Both the cadre nucleocapsid protein (HBcAg) and the 'early' poly peptide (which makes HBeAg) are translated from the C gene. HBcAg is essential for viral packaging and is an integral part of the nucleocapsid. HBeAg is a soluble protein that is not part of the virus particle. Detection of HBeAg in the serum is correlated with viral replication and is a marking for astringent illness. Information technology is most commonly found in those with acute hepatitis B and those with chronic HBV infection with high viral load.[1]
9.2. Clinical features
There is a broad spectrum of clinical affliction with HBV infection, from subclinical through to fulminant hepatitis. Persistent infection can lead to chronic liver disease, potentially causing cirrhosis or hepatocellular carcinoma.
9.2.1. Serological markers of infection
The HBV antigens and their associated antibodies are serological markers of HBV infection or vaccination (Tabular array 9.1). At to the lowest degree one serological marker is present during the unlike phases of infection (Table 9.ii).
| Antigen | Antibody |
|---|---|
| HBsAg | Anti-HBs |
| HBcAg | Anti-HBc |
| HBeAg | Anti-HBe |
| Serological marker | Estimation | |||
|---|---|---|---|---|
| HBsAg | Total | IgM | Anti-HBs | |
| – | – | – | – | Never infected |
| + | – | – | – | Early on astute infection; transient (upwardly to 18 days) after vaccination |
| + | + | + | – | Acute infection |
| – | + | + | + or – | Acute resolving infection |
| – | + | – | + | Recovered from past infection and is immune |
| + | + | – | – | Chronic infectiona |
| – | – | – | + | Immune if ≥x IU/L vaccinated or natural infection |
Central: Anti-HBc = antibody to hepatitis B cadre antigen; anti-HBs = antibody to hepatitis B surface antigen (HBsAg); IgM = immunoglobulin M; + = positive test issue; – = negative test result.
- HBeAg positive (HBeAg+) correlates with high viral load and increased adventure of transmission; HBeAg negative (HBeAg–) correlates with lower viral load and reduced risk of developing cirrhosis or cancer.
Adapted from: Van Damme P, Ward J, Shouval D, et al. 2018. Hepatitis B vaccines. In: Plotkin SA, Orenstein WA, Offit PA (eds). Plotkin's Vaccines (seventh edition). Philadelphia, United states: Elsevier. Table 25.1.
Any difficulties with interpreting serological results for cases and contacts should be discussed with an infectious diseases physician or the laboratory. See the 'Hepatitis B' affiliate of the Communicable Disease Control Manual for recommendations for HBV case and contact management.
9.2.2. Acute hepatitis
The virus preferentially infects liver cells, multiplying in the liver and releasing large amounts of HBsAg, which is present in the blood of people with active infection. The incubation period varies between 45 and 180 days and is commonly 60 to 90 days.
HBV is not directly cytopathic; the host'south immune response leads to decease of infected liver cells. About infected people mount an effective immune response that leads to eradication of infection over a period of several months. Approximately eighty pct of adults with acute infection have symptomatic hepatitis, and the remaining 20 pct can exist asymptomatic (but these proportions vary).[2]
The mutual symptoms of astute hepatitis B disease are fever, jaundice, angst, anorexia, nausea, vomiting, myalgia and abdominal pain. Jaundice usually develops inside two weeks of onset of the disease, and dark urine and/or clay coloured stools might appear up to five days before clinical jaundice. Clinical signs and symptoms of acute hepatitis B usually resolve one to iii months later.[1]
In that location is a small gamble of liver failure (less than one percent) with acute infection; if failure occurs, about half volition dice or require emergency liver transplantation.
nine.two.3. Chronic HBV infection
The primary burden of HBV affliction occurs in people with chronic HBV infection. Chronically infected people are identified past presence and persistence of HBsAg in their serum for at least six months. The historic period of acquisition of HBV is strongly associated with the risk of developing chronic HBV infection. Approximately 90 percent of those infected perinatally or in infancy develop chronic HBV infection, compared with 30 percent of children infected between ages 1 and 4 years and less than v percent of people infected every bit adults.
Infants seldom mount an immune response to HBV infection, and infection in infancy is often asymptomatic. Asymptomatic chronic infection stimulates persistent allowed responses that may eventually lead to cirrhosis (decades later); cirrhosis and chronic infection increase the risk of development of hepatocellular carcinoma.
| Phases of HBV infection | Features |
|---|---|
| Immune tolerance phase | Prolonged flow of active viral replication without active liver disease. Seen in children who larn infection perinatally. |
| Allowed clearance phase | Agile viral replication and active liver affliction |
| Inactive chronic HBV infection | Depression or absent viral replication and remission of active liver affliction |
| Reactivation | HBV replication after inactivity, seen in some patients |
In that location are up to iv phases of chronic infection as in Tabular array 9.iii testify; not all are present in all infections.[one] The initial phase of infection may concluding 10 to 30 years, during which spontaneous clearance rates of HBeAg in the serum are less than one percent per year.
Chronically infected people who are HBsAg positive can also have detectable HBeAg in the serum; this combination is considered most infectious. Although recent evidence suggests HBeAg negative patients are less infectious, information technology is dependent on HBV Dna levels. Whatever the case, both groups can be an ongoing source of infection to susceptible individuals. In the early years of chronic infection, high rates of viral replication are common, and both HBeAg and loftier levels of HBV Dna are present in the blood. In later years, HBeAg may be absent from the blood, and HBV DNA levels (viral load) are usually lower, both of which correspond with lower rates of viral replication.
Information technology is estimated that 4.5 percent of HBsAg positive individuals (12 million people worldwide) have been co-infected with hepatitis D virus (HDV), which is a significant contributor to HBV-associated cirrhosis and hepatocellular carcinoma.[iii] The highest prevalence is seen in those with hepatitis C and HIV, and in certain geographic areas.
ix.2.four. Routes of transmission
HBV is commonly transmitted through contact with infected blood or body fluids during childbirth, contact with broken skin, sexual intercourse or intravenous drug use. Although HBV can exist found in all trunk fluids, blood has the highest concentration and saliva the everyman. HBV in dried blood remains infective for at least ane week.[four]
Perinatal (vertical) transmission
The chief source of HBV infection is perinatal exposure from mothers with chronic HBV infection. Transmission usually occurs at the time of birth. The in utero manual of HBV is relatively rare, bookkeeping for less than two percent of infections transmitted from mother to baby.[5]
If no prophylaxis is given to the babe, the baby of an HBeAg positive mother has a lxx–ninety percent risk of infection, while the baby of an HBeAg negative HBsAg positive carrier mother has a 5–twenty percent risk of infection. Over xc percent of infants who acquire infection perinatally become chronic carriers.
Person-to-person (horizontal) transmission
Not-sexual person-to-person manual probably occurs from inadvertent percutaneous or mucosal contact with blood or infectious body fluids among people in close daily contact (household members).
The main sources of transmission are:
- sexual contact with an infected private
- percutaneous exposure to blood or infectious body fluids
- needle-stick injuries or sharing needles.
Those travelling to loftier endemic countries are at higher run a risk of exposure (come across below).
ix.3. Epidemiology
ix.3.1. Global burden of disease
Approximately two billion people worldwide had been exposed to HBV in 1995. In 2015, based on serological information, around 3.five percentage of the full general population globally were infected with HBV and more than than 250 million people were estimated to take chronic infection and these people remain at risk of developing cirrhosis and hepatocellular carcinoma.[6, 7] More than than 90 percent of individuals with chronic HBV resided in the Asia–Pacific region, where about countries have high prevalence rates of HBV infection (the population rate of HBsAg positivity is betwixt 5 and 20 percent) and more than 99 pct of HBV-infected people in this region caused infection through vertical transmission from their mother (usually at the time of delivery) or in early childhood.[eight] As an example of this risk, 22.8 million out of lxxx meg people living in China with chronic HBV infection are women of changeable age.[9] Acquisition of HBV during adulthood (usually via sexual transmission or injecting drug utilize) is associated with a high rate of symptomatic hepatitis merely a low charge per unit of chronic infection.
The introduction of universal childhood HBV immunisation has inverse the epidemiology of chronic infection in many countries, simply it will exist several decades (one to two human being generations) before the total benefits are realised. In Mainland china, for example, within xx years since the introduction of HBV immunisation, mother-to-child transmission has been cut by 97 percent; 120 million new HBV infections and 28 million chronic infections accept been averted.[9] Thirty years afterwards the introduction of a HepB immunisation programme for newborns in Taiwan, infant fulminant hepatitis bloodshed and, in those anile five to 29 years, chronic liver affliction and hepatocellular carcinoma mortality had all decreased past more than than 90 percent.[10]
The earth tin can be divided into regions with high (8 percentage and over), high-moderate (5–7 per centum), low-moderate (2–four pct) and low (less than ii percent) prevalence of chronic infection, defined equally the presence of HBsAg in serum.[eleven, 12] In regions with a high prevalence of chronic infection, the lifetime risk of exposure to HBV is most 80 per centum, with nigh infections occurring in the first decade of life. The Pacific Islands and most of Asia (except Nihon and India) are high-prevalence regions. Other loftier-prevalence regions include Sub-Saharan Africa and Latin America.[12] In dissimilarity, in countries with a low HBsAg prevalence, the lifetime gamble of HBV exposure is less than 20 pct, with most infections caused in adulthood. New Zealand has a low overall prevalence of hepatitis B railroad vehicle but contains certain populations with loftier prevalence (see section ix.3.2 below).
9.3.ii. New Zealand epidemiology
Before the introduction of HBV immunisation in New Zealand, HBV transmission was mutual among preschool and schoolhouse-aged children. The exact mode of transmission is uncertain, merely is thought to exist related to shut contact. In the eastern Bay of Enough region nearly one-half of the population were infected past age 15 years.[13, 14] Even after the introduction of universal HepB in 1988 (run across Appendix 1), there were regions in New Zealand where children were still at take a chance of HBV infection due to poor immunisation coverage rates.[15, 16, 17]
Acute HBV infection
Simply acute hepatitis B is a notifiable disease in New Zealand; notification rates exercise not describe the burden of chronic HBV infections.
The HBV notification rate in 2019 was 0.6 per 100,000 population (28 cases), like to the 2018 rate (0.7 per 100,000, 33 cases). The highest notification charge per unit was in the 30–39 years age group (1.1 per 100,000) followed by 50–59 years and lxx years and over (both 0.8 per 100,000). The notification rate was higher for males (0.nine per 100,000) than for females (0.3 per 100,000) (ESR, viii June 2020).
Ethnicity was recorded for all cases. The Māori (1.three per 100,000) ethnic grouping had the highest hepatitis B notification rate followed by the Asian (0.seven per 100,000) ethnic group.
The nearly common reported chance factors were overseas travel, migration and sexual contact with a confirmed case or carrier.
Hepatitis B notifications accept declined from 609 cases in 1984 to 28 cases in 2019 (see Figure 9.1). While hard to quantify accurately, the introduction of universal baby immunisation in 1988 has contributed to the dramatic refuse in the number of newly notified cases of HBV infection.
Figure 9.ane: Notifications of hepatitis B, 1997–2019

Source: ESR
For recent data on acute hepatitis B notifications, refer to the almost contempo notifiable illness annual reports from ESR (available on the Public Health Surveillance website).
Chronic HBV infection
The Hepatitis Foundation of New Zealand reports that around 120,000 people in New Zealand are living with chronic HBV infection, around 50 percent are diagnosed, merely merely around seven,000 are beingness treated. Based on 2016 data, around 1,000 new cases are diagnosed each year nationally. Mathematical modelling anticipated that the prevalence of chronic HBV infection would drop from 3.3 percentage in 2016 to 2.four percent by 2030, falling short of the elimination targets prepare by the WHO in 2016.[xviii]
The National Hepatitis B Screening Programme plant that between 1999 and 2002 in the N Isle, the highest rates of chronic HBV infection were amongst Chinese (9.1 percentage), Pacific peoples (8.5 percentage) and Māori (five.viii percent). Although Europeans were not specifically targeted in this screening programme, they accept an estimated prevalence rate of 1 percent (college than in Australia, N America and Europe), reflecting an increased run a risk of babyhood horizontal transmission.[19]
A New Zealand-based modelling study estimated that until the year 2100, people with chronic HBV infection will continue to provide a source of infection to susceptible people.[20] Increased immigration from high-prevalence countries in the Asia–Pacific region is likewise likely to influence HBV prevalence in New Zealand.
Because people who acquire chronic HBV infection in childhood usually do non develop hepatocellular carcinoma until aged forty years or older, the introduction of a universal HBV vaccination in 1988 is unlikely to take a meaning effect on the incidence of hepatocellular carcinoma until approximately 2030.
A retrospective laboratory data study of antenatal HBsAg tests from the Midlands region (Bay of Plenty, Eastern Bay of Plenty, Waikato and Rotorua) between 1997 and 2009 institute a failing prevalence of HBV infection. This subtract was seen across all age groups, but was most marked in antenatal tests of women aged under 20 years, due to receipt of funded HepB in childhood.[21]
A long-term follow-upwards study in New Zealand showed that horizontally acquired HBV infection during childhood in Māori and Pacific peoples correlates with increased rates of hepatocellular carcinoma and liver-related mortality.[22] This study emphasises the importance of early protection of the infant with vaccination.
Strategy for prevention
In 1988 New Zealand was one of the first countries to innovate universal baby hepatitis B immunisation. As of 31 December 2019, 93 percent of New Zealand children aged two years had completed a primary grade of HepB, which confers lifelong immunity in approximately 95 percentage of those vaccinated.
nine.four. Vaccines
9.4.i. Available vaccines
The specific monovalent and combination HepB vaccines licensed (canonical for use) and available (marketed) in New Zealand contain recombinant HBsAg (HepB).
Funded vaccines
- HepB (Engerix-B, GSK): contains xx µg HBsAg per dose; information technology does not contain a preservative. Other components and residuals include aluminium hydroxide, sodium chloride, sodium phosphate dehydrate, sodium dihydrogen phosphate and traces of polysorbate 80.
- DTaP-IPV-HepB/Hib (Infanrix-hexa, GSK): diphtheria, tetanus, acellular pertussis, inactivated polio, hepatitis B and Haemophilus influenzae blazon b vaccine (see section 6.4.1 for more information).
Other vaccines
HepA-HepB (hepatitis A and hepatitis B vaccine): Twinrix and Twinrix Junior (GSK) (see as well section 9.four.1).
9.4.2. Efficacy and effectiveness
Clinical trials in high-hazard groups accept shown a vaccine efficacy of 85–95 percent for HepB vaccines.[eight]
See also section xv.4.ii for information about the DTaP-IPV-HepB/Hib vaccine.
Immunogenicity
Serum anti-HBs antibody ≥10 IU/L, measured 1–two months after immunisation, is considered by WHO as a correlate of long-term protection.[8] In the principal intendance setting, individuals who have had a documented seroconversion after three injections are expected to take lifelong immunity with no need for further boosters, even if circulating antibody is later not detectable.
Smoking, obesity, HIV infection and chronic disease (including renal failure) all reduce vaccine efficacy, but age is the primary factor affecting the response. At to the lowest degree 98 percent of infants, 95 percentage of children and 90 percent of adolescents develop protective levels of antibody afterwards three doses of vaccine. Some non-responders volition not produce acceptable antibody levels to the initial vaccination course, but most reply to further vaccine doses.
However, some people are persistent non-responders. Persistent not-responders often have an dumb immune organisation, such as organ transplant recipients and those with HIV infection or chronic disease, including avant-garde cirrhosis, renal failure or those undergoing haemodialysis. A modest percent (approximately ii–3 pct) of the immunocompetent population may as well fail to arm-twist an antibody response. Loftier-adventure individuals who fail to respond adequately are recommended further vaccinations (see department 9.5.7).
Effectiveness of birth dose given to babies built-in to HBsAg‑positive mothers
Infants vaccinated at birth built-in to infected mothers were three.five times less likely to exist infected with HBV than those who did not receive a nascence vaccination.[eight, 23] For babies of HBeAg-positive mothers, controlled trials have shown that vaccine at birth provides 75 percent protection from infection, while assistants of HBIG in add-on to vaccination provides 85–95 percent protection confronting transmission.[23, 24] Protection is reduced to less than 80 pct when the mother's HBV DNA level (viral load) is greater than 108 IU/mL (or 108 copies/mL).[25] In this situation, administration of tenofovir (an antiviral agent) to the mother during the last trimester is recommended and funded.
Duration of immunity
The evolution of anti-HBs antibodies subsequently a primary vaccination course (three injections and seroconversion) indicates development of immune memory. The quantity of antibody in serum is thought to determine the length of time the antibiotic titre can be detected in the claret, although any reading ≥10 IU/L post-vaccination course is considered protective.[25, 26] Children who are given booster doses upwardly to 12 years subsequently the primary series show strong anamnestic (secondary) responses, indicating that booster is unnecessary one time a seroprotective level is reached later the three-dose primary vaccination course.[26, 27]
Long-term protection from clinical infection, despite loss of detectable neutralising antibiotic, is thought to reverberate a potent cellular memory immune response following HBV vaccination.[28] Even though a large proportion of vaccine recipients may have undetectable antibody within 7 years of vaccination, at that place is bear witness from Germany,[28] Taiwan,[29] Alaska[xxx] and Hawaii[31] that boosters of HepB are unnecessary post-obit completion of infant immunisation.
Sustained allowed memory, including circulating retention B and T cells, and long-term protection take been shown 20–xxx years later on complete master immunisation of allowed competent adults in the absence of natural or artificial boosting.[24]
In full general, vaccine recipients who are subsequently infected with HBV do not develop clinical illness but may take anti-HBc present in plasma.[1]
Impact on chronic HBV infection
In all populations, where it has been measured, immunisation has led to a dramatic drop in HBV chronic infection.[31] For instance, chronic HBV infection dropped from 16 percent to zero in Alaska equally a consequence of 96 percent immunisation coverage. In Taiwan, the incidence of hepatocellular carcinoma likewise decreased equally a result of the immunisation plan in children.[33, 34] Adolescents and adults who were offered universal HBV vaccination in infancy had more than 75 per centum lower prevalence of HBV infection and anti-HBc prevalence than those for whom immunisation was unavailable.[35]
9.iv.three. Transport, storage and handling
Transport according to the National Standards for Vaccine Storage and Transportation for Immunisation Providers 2017 (2nd edition).
Store at +2°C to +8°C. Practice not freeze. DTaP-IPV-HepB/Hib and HepB vaccines should be stored in the night.
DTaP-IPV-HepB/Hib (Infanrix-hexa) must be reconstituted past adding the entire contents of the supplied container of the DTaP‑IPV-HepB vaccine to the vial containing the Hib-PRP pellet. Later adding the vaccine to the pellet, the mixture should exist shaken until the pellet is completely dissolved. Use the reconstituted vaccine every bit before long as possible. If storage is necessary, the reconstituted vaccine may exist kept for up to eight hours at 21°C.
nine.4.iv. Dosage and administration
DTaP-IPV-HepB/Hib
Each 0.5 mL dose of DTaP-IPV-HepB/Hib (Infanrix-hexa) vaccine contains ten μg of HBsAg, and is administered by intramuscular injection (run into department 2.2.3).
HepB
The dose of HepB vaccine varies according to the vaccine manufacturer, the age of the individual and/or their health condition (meet section 9.v for recommendations):
- Engerix-B 20 μg (GSK): 20 μg HBsAg per 1.0 mL
- Engerix-B paediatric x μg (GSK): 10 μg per 0.5 mL.
HepB vaccine is administered by intramuscular injection. It can be besides administered by subcutaneous injection, if indicated for bleeding disorders (see department 2.2.3).
Co-administration with other vaccines
Hepatitis B vaccines may be given at the same time as all other vaccines on the Schedule, including measles, mumps and rubella (MMR) vaccine.
If a form of vaccine is interrupted, it may be resumed without repeating prior doses (see Appendix 2).
ix.5. Recommended immunisation schedule
| Recommended and funded |
|---|
| Household or sexual contacts of HBsAg-positive patients (ie, patients with acute or chronic HBV infection) |
| Babies of HBsAg-positive mothers (ie, mothers with acute or chronic HBV infection) – require a nascence dose plus the three-dose main series (HBIG is also given to these babies at birth) |
| Children and adolescents aged under xviii years who are considered non to have accomplished a positive serology past one month later on vaccination and crave additional vaccination or require a primary class of vaccinationa |
| Individuals who are HIV-positiveb |
| Individuals who are hepatitis C-positivec |
| Following not-consensual sexual intercourse |
| Prior to planned or following immunosuppressionb,d |
| Prior to or following solid organ transplantb,d |
| Individuals post-HSCTb |
| Following needle-stick injury |
| Patients on dialysisb,d |
| Recommended, not funded |
| Adults at occupational risk (run across section 4.8) |
| Adults at risk of infection past sexual exposure:
|
| Individuals with haemophilia and other regular recipients of claret products |
| Prison inmates |
| Current or recent injecting drug users |
| Individuals with developmental disabilities |
| Migrants from HBV endemic countries (HBsAg prevalence ≥two percent)f |
| Travellers to HBV endemic regions (HBsAg prevalence ≥2 percent)f |
- Serological testing is not routinely recommended, see Figure 9.iii.
- See also section 4.3.3.
- Hepatitis C patients should besides receive hepatitis A vaccine, although this is not currently funded.
- The menstruation of immunosuppression due to steroid or other immunosuppressive therapy must be longer than 28 days.
- twoscore µg of HepB is recommended for adult dialysis patients or for adult liver or kidney transplant patients.[36] See Table ix.5.
- See the Centers for Disease Control and Prevention website for countries with an HBsAg prevalence ≥two percent. Consider combined Hep A and B vaccination for travellers to these regions.
9.5.1. Usual childhood schedule
A master course of hepatitis B vaccination is given every bit iii doses of DTaP-IPV-HepB/Hib at ages 6 weeks, 3 months and 5 months (Table 9.5). If a course of immunisation is interrupted for any reason, it may exist resumed without repeating prior doses (see department 9.5.3 and Appendix ii).
| Age | Vaccine | Comment |
|---|---|---|
| 6 weeks | DTaP-IPV-HepB/Hib | Master serial |
| 3 months | DTaP-IPV-HepB/Hib | Primary series |
| 5 months | DTaP-IPV-HepB/Hib | Primary serial |
Preterm infants of HBsAg-negative women
Some depression birthweight or preterm infants may have a reduced response to HepB vaccine at nativity.[37] However, by the chronological age of 1 month, all medically stable preterm infants, regardless of initial birthweight or gestational age, answer to HepB as well equally term and larger infants.[38] Because New Zealand'southward Schedule starts at historic period 6 weeks, low birthweight and preterm infants are expected to respond to HepB content in the DTaP-IPV-HepB/Hib vaccine. (Meet also sections 4.two.1 and four.two.2.)
Infants with liver or renal illness
HepB vaccine is funded for liver or kidney transplant patients and for dialysis patients. For infants requiring transplants, see 'Solid organ transplantation' in section 4.3.ten. For infants undergoing dialysis, see 'Chronic kidney affliction' in section 4.iv.
nine.5.2. Infants born to HBsAg-positive mothers
The routine schedule for these infants is a nascency dose of monovalent HepB plus HBIG, then three routine doses of DTaP-IPV-HepB/Hib at ages six weeks, 3 months and 5 months.
All pregnant women should receive antenatal screening for hepatitis B infection by testing for HBsAg. Infants of HBsAg-positive mothers are to be notified at birth using the form HE1446: Consent for hepatitis B vaccine and hepatitis B immunoglobulin and notification to the Medical Officer of Wellness, available from the Health Ed website or the local authorised health education resource provider or public health unit of measurement.
Infants born to HBsAg-positive mothers should receive:
- 100–110 IU HBIG neonatal, at or as close as possible to birth
- a nascence dose of HepB which should be given at or equally close as possible to birth (preferably within 12 hours).
If HBIG and/or HepB is inadvertently omitted, administrate as soon as the omission is recognised. HBIG can be administered up to seven days postal service-delivery. If there is a filibuster for longer than seven days, seek specialist advice.
These infants should then continue as per the Schedule at ages half dozen weeks, 3 months and five months. Serological testing is required at 9 months of age (see beneath).
The vitamin Yard injection may also be given at the same time, in the aforementioned limb as the HBIG, only non at the aforementioned site.
Occasionally women have not been tested for their HBsAg status during the antenatal period. If a adult female's HBsAg condition is unknown at the time of delivery, the baby should be given HepB at the time of delivery while waiting for the result of an urgent HBsAg examination on the mother. If she is institute to be HBsAg positive, the infant should be given HBIG every bit soon as possible, upwardly to vii days post-delivery. Immunoprophylaxis is most constructive when given within 12 hours of delivery.[38] Subsequent vaccine doses are given as per the Schedule.
It is essential to take blood to decide whether the babe has seroconverted (anti-HBs positive) or has become infected despite immunoprophylaxis (HBsAg positive), or is neither infected nor immune (ie, HBsAg negative and anti-HBs negative). Testing should be performed at 9 months of age to avoid detection of anti-HBs from HBIG administered during infancy and to maximise the likelihood of detecting tardily onset HBV infections.[38] Infants of HBsAg-positive mothers should exist placed on a practice recall system to have their blood tested at ix months of age. Bank check at the 12-month immunisation result to ensure that testing has occurred. The serology results should be interpreted as in Figure ix.2.
| Screen all women in early pregnancy for hepatitis B wagon | |
|---|---|
| Woman is HBsAg positive No | |
| Yep | |
| All HBsAg-positive pregnant women should also be tested for HBeAg and should accept HBV DNA measured. The results should be discussed with a specialist or the woman should immediately be referred to a specialist for ongoing care. Requite the baby hepatitis B protection as follows. | |
| At age | Action to be taken |
| Birth | Requite HBIG 100–110 IU and HepB |
| half-dozen weeks | DTaP-IPV-HepB/Hib |
| 3 months | DTaP-IPV-HepB/Hib |
| 5 months | DTaP-IPV-HepB/Hib |
| ix months | Have a blood test to bank check for hepatitis B infection (HBsAg) and for vaccine-induced immunity (anti-HBs). If HBsAg is negative and anti-HBs level is ≥10 IU/50 at historic period 9 months, immunity is proven. If HBsAg is positive, the infant has become infected despite prophylaxis: refer to an advisable specialist. If HBsAg is negative and anti-HBs level is <10 IU/L at age 9 months, requite a farther 3 doses of HepB at least 4 weeks apart. Recheck serology 4 weeks after the last dose. If there is no seroconversion after the third further dose of HepB (ie, if anti‑HBs is still <10 IU/L), discuss with a specialist. |
| All other vaccines should exist administered as per the Schedule. | |
Neonatal HBIG plus vaccine will fail to prevent vertical HBV transmission in up to 20 percentage of infants born to HBsAg-positive mothers with serum HBV DNA levels greater than 108 IU/mL (or 2 x107 copies/mL). These mothers are usually young, with normal alanine transaminase, and are HBeAg-positive. If the mother's HBV Dna level is greater than 200,000 IU/mL[25, 39, 40], administration of tenofovir (an antiviral agent) during the last trimester is funded.
The number of such high-risk pregnancies appears to exist increasing in this country as a result of the immigration of young Asian women of childbearing age, of whom approximately 8 pct are HBsAg-positive with the majority of those also HBeAg-positive. In contrast, the number of HBsAg-positive Māori and Pacific women of childbearing age has decreased markedly due to infant vaccination. In addition, most HBsAg‑positive Māori and Pacific women are HBeAg-negative, with lower HBV DNA levels (below 10eight IU/mL).
Infants born to mothers who received oral antiviral therapy for chronic HBV must nevertheless receive the recommended neonatal HBIG/vaccine schedule. All other vaccines are administered every bit per the Schedule.
See Appendix 6 and section 9.8.1 for more information about passive immunisation and HBIG.
Preterm and low birthweight infants of HBsAg-positive women
Preterm and low birthweight infants of HBsAg-positive women should be managed as above, regardless of birthweight or gestation.
9.5.3. Catch-ups for children and adolescents
HepB is recommended and funded for everyone aged under 18 years. If HepB is non given during the first year of life, iii doses of vaccine are recommended.
For adolescents aged 11–15 years, an alternative 2-dose hepatitis B catch-upwardly schedule may exist considered using the monovalent HepB with the second dose given four to six months later the get-go.
Meet Appendix 2 for catch-up schedules.
Children and adolescents with liver or kidney affliction
HepB vaccine is funded for liver or kidney transplant patients (recommend six months post-transplant) and for dialysis patients.
Run across Effigy 9.3 and Effigy ix.4 for serological testing and vaccination recommendations. If non-immune, children aged under 16 years should receive three doses of HepB; those aged sixteen years and older should receive iv doses of HepB given at 0, 1, 2 and 12 months. If in that location is an inadequate allowed response to the initial iii-dose HepB series (run into Figure 9.4), give a further three doses, every bit appropriate for historic period.
See also 'Solid organ transplantation' in section 4.iii.10, 'Chronic kidney disease' in section 4.four and 'Chronic liver disease' in section iv.5.
9.5.4. Eligible adults aged xviii years and older
| Who | Vaccine | Dose | Volume (mL) | Number of doses | Schedule |
|---|---|---|---|---|---|
| Dialysis patients, liver or kidney transplant patients | HepB | 40 µg | 1.0 | 3 | 0, one and six months |
| HIV patients | HepB | 20 µg | 1.0 | 4 | 0, ane, ii and 12 months |
| Other eligible adults (encounter Tabular array 9.4) | HepB | 20 µg | 1.0 | three | 0, 1 and vi months |
Adult dialysis or adult liver or kidney transplant patients
These adults may accept a reduced response to HepB,[36, 41] then 3 higher doses (40 µg per dose) are recommended and funded.
Come across section nine.5.7 for information almost mail service-vaccination serology.
(See also 'Solid organ transplantation' in section four.3.x and recommendations provided in IMAC factsheet 'Immunisation for adults pre-dialysis, on dialysis or pre-/post-kidney transplant' available on the IMAC website.)
Developed HIV patients
Developed HIV patients should receive 4 doses of HepB (20 µg per dose) at 0, i, 2 and 12 months.
(See also 'HIV infection' in section 4.3.12 and recommendations provided in IMAC factsheet Immunisation for adults with HIV infection available on the IMAC website.)
Other eligible adults
The optimal dosing regime is iii doses of xx µg HepB given at 0, ane and half-dozen months. See the manufacturer'southward data canvas for sub-optimal accelerated HepB schedules if dosing is time constrained. For other eligible adults, run into Table four.eight, 'Other special groups' in department four.vi.
9.5.5. Pregnancy and breastfeeding
HepB may be given during pregnancy and while breastfeeding. Acute HBV infection in pregnant women may consequence in severe acute hepatitis for the mother, with associated increased chance of fetal loss or neonatal infection. Vaccination should not exist withheld from a susceptible meaning woman at increased risk of acquiring hepatitis B (eg, the sexual partner of an injecting drug user, or known infected male).
9.5.6. (Re)vaccination
Hepatitis B-containing vaccines are funded for vaccination and revaccination of eligible children, equally follows. See besides sections four.2 and 4.3.
DTaP-IPV-HepB/Hib (Infanrix-hexa)
An additional iv doses (equally appropriate) of DTaP-IPV-HepB/Hib are funded for vaccination or revaccination of children aged under 10 years:
- post-HSCT or chemotherapy
- pre- or post-splenectomy
- pre- or post-solid organ transplant
- undergoing renal dialysis
- prior to planned or following other severely immunosuppressive regimens.
Up to five doses of DTaP-IPV-HepB/Hib are funded for children aged nether x years receiving solid organ transplantation.
Monovalent HepB
HepB is funded for children anile under 18 years who are considered not to accept achieved a positive serology and crave additional vaccination.
9.five.7. Serological testing
- Serological testing is non routinely recommended – immunisation is highly effective.
- Almost people with documented prove of iii HepB vaccinations volition exist allowed for life.
- Unnecessary testing leads to unnecessary actress vaccination.
- Infants born to HBsAg-positive mothers and some individuals who require protection in relation to their employment (eg, health intendance professionals) crave post-vaccination serology.
- Where in that location is concern nearly amnesty follow Figure 9.three.
Screening for chronic infection
Screening for the antigen (HBsAg) is useful where there is increased likelihood of the individual already being infected.
The Hepatitis Foundation of New Zealand recommends that the post-obit individuals are well-nigh at risk of HBV:[42]
- people of Māori, Pacific or Asian ethnicity, unless fully vaccinated with HepB vaccine every bit an infant
- people born in an area of high hepatitis B endemicity, including Asia, the Pacific Islands, Africa, the Middle East, southern Europe or the northern or eastern parts of New Zealand'due south North Island
- people built-in to a female parent or who take a close family member who has chronic HBV infection
- people who live with someone who has HBV
- people who take had unprotected sexual contact with an HBV-infected person
- people who have e'er injected drugs
- people who have received a tattoo using unsterile equipment.
Screening for HBsAg is too part of routine antenatal intendance (see section 9.five.2).
All HBsAg-positive individuals should exist offered follow-up nether the Hepatitis Foundation Hepatitis B Follow-upward Plan to enable early diagnosis and treatment of the complications of severe liver affliction and hepatocellular carcinoma. Vaccination is funded for household or sexual contacts of HBsAg-positive people (ie, contacts of people with acute or chronic HBV infection).
To ostend chronic HBV status, repeat testing after 6 months and if withal positive, refer patient to The Hepatitis Foundation.
Serological testing for high-gamble groups
Serological testing is not routinely recommended – immunisation is highly effective.
- Serological testing is only indicated in loftier-chance groups (run into Table 9.vii). These high-risk groups are at higher risk of exposure to HBV, at higher adventure of having astringent disease or are more than susceptible to disease.
- A flow diagram (Figure 9.3) is included to assist in deciding whether pre- and/or post-vaccination serological testing is indicated. Figure 9.3 may be used for any individual aged 12 months or older, such every bit for the direction of claret and body fluid exposures, or when an developed presents to primary intendance.
| Household or sexual contacts of HBsAg-positive patients (ie, patients with acute or chronic HBV infection) |
| Electric current or recent injecting drug users |
| Individuals who change sexual partners frequently (eg, sexual activity workers) |
| Immunocompromised individuals, including HIV-positive patients |
| Following non-consensual sexual intercourse |
| Individuals prior to planned immunosuppressive therapies for 28 days or more |
| Individuals following immunosuppressive therapies for 28 days or more |
| Solid organ and mail-HSCT patients |
| Post-obit percutaneous injury (eg, needle-stick injury) |
| Adults at occupational-related risk (run into department 4.8) |
| Individuals with haemophilia and other regular recipients of blood products |
| Inmates of custodial institutions |
| Individuals with developmental disabilities |
| People with chronic illness (eg, chronic renal failure requiring haemodialysis, or chronic liver disease) |
| Migrants from HBV endemic regions (where HBsAg prevalence is ≥2 percent)* |
* Run into the Centers for Disease Control and Prevention website for countries with an HBsAg prevalence ≥2%. Consider combined Hep A and B vaccination for individuals travelling to these regions.
Figure 9.3: Period diagram for serological testing for hepatitis B
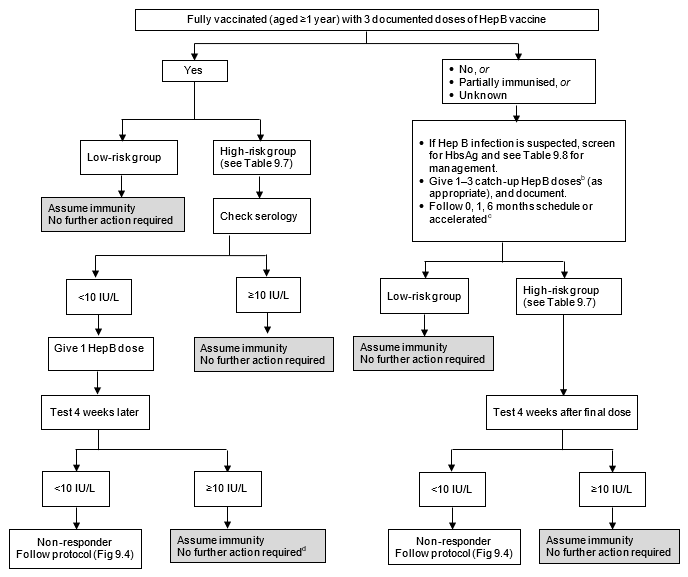
- HBIG may exist recommended for non-immune individuals. See Tabular array ix.8.
- Practice non count any nascence doses of HepB vaccine. Encounter Table ix.4 for the list of funded weather for HepB vaccine.
- Meet the manufacturer's data sheet for accelerated HepB schedules.
- Haemodialysis patients need annual testing and boosting if required.
The non-responder protocol
Virtually vaccine recipients will develop a high anti-HBs titre, usually greater than 100 IU/L, which ordinarily wanes over time.
Fully vaccinated individuals (ie, those who have received three documented doses of HepB) who have at any time had anti-HBs ≥10 IU/50 do not need any booster doses, even if antibodies afterward wane to undetectable levels, which occurs in nearly individuals by seven years after the last vaccination. Adults accept been shown to take an anamnestic antibody and cellular immune response to a HepB dose given 20–30 years after the principal immunisation.[24] If exposed to HBV, they will have a secondary anamnestic allowed response that will prevent replication of the virus.[1, 43]
Note: Some laboratories may require a higher anti-HBs antibody level for proof of amnesty. Delight follow the testing laboratory's interpretative comments.
If a high-run a risk individual does not reach a titre of ≥10 IU/L by 4 weeks following one HepB dose, they should be considered a not-responder and follow the non-responder protocol (Figure 9.4).
Figure 9.4: The non-responder protocol
Private is loftier-take a chance (see Tabular array nine.7), has received three documented doses of HepB plus a booster dose and has an anti-HBs <ten IU/50:
- Complete a 2nd course of three HepB vaccine doses.
- Repeat the serology four weeks after the terminal HepB vaccine dose.
- If anti-HBs ≥10 IU/L, assume immunity. No further action is required.
- A third course of iii doses of 40µg HepB vaccine (ie, ii injections per visit of Engerix-B xx µg) is brash for high run a risk individuals who neglect to respond to a second course.[44] Alternatively, in that location is also some testify that using a double dose of HAV-HepB (Twinrix) at 0, 1 and 6 months can right this hyporesponsiveness, using the bystander carrier outcome of the HAV component,[45] but this is not funded.
- Repeat serology 4 weeks after concluding HepB dose.
- If, after the tertiary course of three HepB vaccine, a person has not achieved anti-HBs ≥10 IU/L, they should be considered a persistent non-responder to vaccination.
- Persistent not-responders with no immunocompromise who accept completed the primary series and farther courses of three vaccine doses should be monitored for wild-type disease, but literature reports disease from vaccine failures are rare. They should be considered 'unprotected' against hepatitis B and advised to minimise the chance of exposures. Parenteral or mucosal exposure to HBV requires HBIG within 72 hours.
Intradermal injections to correct this hypo-responsiveness have been used in the past, but they are technically hard and not recommended.
9.6. Contraindications and precautions
Meet besides department two.1.3 for pre-vaccination screening guidelines and section ii.1.4 for general contraindications for all vaccines.
The just specific contraindication to HepB is anaphylaxis following a previous dose, or individuals with a history of allergic reactions to yeast or whatsoever of the vaccine'southward components. Immunisation of previously infected subjects is wasteful, but not harmful.
See section xv.6 for contraindications and precautions to DTaP‑IPV‑HepB/Hib vaccine.
9.vii. Potential responses and AEFIs
Run into department 15.7 for potential responses and AEFIs with DTaP‑IPV‑HepB/Hib vaccine.
9.7.1. Potential responses
Small-scale side-furnishings – including local tenderness and redness, nausea, diarrhoea, full general malaise and fever – are more common in adults than in children and, except for local reactions, occur at rates shut to those seen with a placebo. Minor reactions reported after receiving the vaccine include a temperature >37.seven°C in one–6 percent; pain in 3–29 percent; and erythema, headache or swelling in 3 pct of vaccine recipients.
9.vii.two. AEFIs
Allergic reactions have been reported merely are rare. Anaphylaxis following vaccination is extremely rare (estimated to be i.1 cases per 1000000 doses).[46]
A number of studies have examined and failed to find disease events linked to hepatitis B immunisation.[47] These studies have documented no increased risk of multiple sclerosis,[48, 49, 50] diabetes, chronic fatigue syndrome,[51] encephalomyelitis or hair loss.[52] Rarely, transient thrombocytopenia[53] and myalgia and arthralgia[54, 55] have been reported later HepB vaccination.
9.eight. Public health measures
The elimination of HBV manual is at present a realistic public health goal[7, 56] especially with the proven effectiveness and condom record of HepB.[57] Achievement of this goal is being facilitated by the implementation of triple emptying strategies in the Western Pacific Region to prevent mother-to-child manual of HIV, HBV and syphilis,[eight] and the employ of nascency dosing HepB regimes, either universally or equally directed.
It is important to ensure vaccination programmes are maintained for the at-adventure populations, especially babies of mothers with chronic hepatitis B infection.
Information technology is a legal requirement that all cases of acute hepatitis B infection be notified to the local medical officer of health.
Babies born to HBsAg-positive mothers should be notified at nativity. The prevention of perinatal manual is covered in department nine.5.ii.
9.8.1. Passive immunisation
HBIG is prepared from donated blood plasma and contains high levels of anti-HBs antibiotic (see Appendix 6). It is given after exposure to HBV and provides passive anti-HBs antibody protection against astute and chronic HBV illness. HBIG prophylaxis should be given in combination with the HepB to confer both passive and active immunity after exposure.
The efficacy of HBIG alone in preventing clinical hepatitis B infection is about 75 percent in adults, merely the protection lasts but for a few months.[1]
Whenever immediate protection is required, immunisation with a vaccine should be combined with simultaneous administration of HBIG at a different site. It has been shown that passive immunisation with HBIG does not suppress the active immune response to vaccination. A single dose of HBIG is sufficient (usually 400 IU for adults, 100–110 IU for newborns).[58] If infection has already occurred at the time of the first immunisation, virus replication is unlikely to exist inhibited completely, but astringent illness and, more chiefly, the development of chronic HBV infection may exist prevented, particularly in the infants of HBsAg-positive mothers.
The management of contacts is summarised in Tabular array ix.8.
| Contact | Serological testing of contact | Immunoglobulin | Immunisation |
|---|---|---|---|
| Any sexual contact, including protected sex | Yep | Aye, immediately afterward claret taken | Yes, immediately after blood taken |
| Household, mucosal or percutaneous | Aye | Yep, if serology negative | Yes, if serology negative |
| Other | Yes | No | Yes, if serology negative |
Source: Ministry building of Wellness. 2012. Communicable Disease Control Manual. Wellington: Ministry of Health (accessed thirty June 2020).
For more than details on control measures, refer to the 'Hepatitis B' affiliate of the Communicable Illness Control Transmission.
9.9. Variations from the vaccine data sheet
See section 15.9 for variations from the DTaP-IPV-HepB/Hib (Infanrix‑hexa) data sheet.
Ministry of Health advises that two doses Engerix-B 20 µg, given four to six months apart, may be given to adolescents aged xi–fifteen years. The manufacturer's information sheet recommends three doses of 10 µg given at 0, ane and 6 months, but in circumstances where compliance may not exist assured, giving xx µg per dose increases the proportion of recipients protected after the first and second doses.
Although the Ministry building of Health and the data canvas recommend 0.5 ml Engerix-B x µg (paediatric conception) for neonates born to HBV infected mothers, where the paediatric presentation is unavailable, the information sheet advises that Engerix-B twenty µg tin can be given to children from birth up to the age of x years.
The Ministry building of Health recommends giving iii doses of 40 ug HepB (ie, 2 doses of Engerix-B 20 µg per visit) to be given 0, 1, and 6 months for adult renal dialysis patients, liver or kidney transplant patients. For adults with HIV, four doses of Engerix-B 20 µg is recommended given at 0, 1, 2 and 12 months. The data sheet advises iv doses of xl µg (ie, two doses for Engerix-B 20 µg per visit) given at 0, one, ii and 6 months for chronic haemodialysis patients and other individuals who have an impairment of their immune system.
References
- Van Damme P, Ward J, Shouval D, et al. 2018. Hepatitis B Vaccines. In Plotkin S, Orenstein W, Offit P, et al (eds) Plotkin'south Vaccines (7th edition), Elsevier: Philadelphia, The states.
- McMahon BJ, Alward WL, Hall DB, et al. 1985. Acute hepatitis B virus infection: relation of age to the clinical expression of disease and subsequent evolution of the carrier state. Journal of Infectious Diseases 151(4): 599–603.
- Stockdale AJ, Kreuels B, Henrion MYR, et al. 2020. The global prevalence of hepatitis D virus infection: systematic review and meta-analysis. Journal of Hepatology.
- Bond WW, Favero MS, Petersen NJ, et al. 1981. Survival of hepatitis B virus later on drying and storage for ane week. Lancet ane(8219): 550–i.
- Modify HJ. 2012. To have B or non to take B: vaccine and the potential eradication of hepatitis B. Periodical of Hepatology 57(four): 715–7.
- Papastergiou V, Lombardi R, MacDonald D, et al. 2015. Global epidemiology of hepatitis B Virus (HBV) infection. Current Hepatology Reports 14(iii): 171–8.
- Globe Health Arrangement. 2016. WHO Global Health Sector Strategy on Viral Hepatitis 2016–2021 (ed). Geneva, Switzerland: World Health Organization. URL: http://apps.who.int/iris/bitstream/10665/246177/one/WHO-HIV-2016.06-eng.pdf?ua=1 (accessed 17 January 2020).
- World Wellness Organization. 2017. Hepatitis B vaccines: WHO position paper – July 2017. Weekly Epidemiological Record 92(27): 369–92.
- World Wellness System. 2019. China steers towards zip new hepatitis B infections (press release). World Health Organization. 30 March. URL: https://www.who.int/hepatitis/news-events/mainland china-hbv-childhood-vaccination/en/ (accessed 3 July 2020).
- Chiang CJ, Yang YW, Y'all SL, et al. 2013. Xxx-year outcomes of the national hepatitis B immunization program in Taiwan. JAMA 310(9): 974–half dozen.
- Harris AM. 2020. Hepatitis B. In CDC 2020 Yellow Book. Wellness Information for International Travel. New York, New York. URL: https://wwwnc.cdc.gov/travel/yellowbook/2020/travel-related-infectious-diseases/hepatitis-b (accessed three July 2020).
- Ott JJ, Stevens GA, Groeger J, et al. 2012. Global epidemiology of hepatitis B virus infection: new estimates of age-specific HBsAg seroprevalence and endemicity. Vaccine 30(12): 2212–9.
- Milne A, Allwood GK, Moyes CD, et al. 1985. Prevalence of hepatitis B infections in a multiracial New Zealand community. New Zealand Medical Journal 98(782): 529–32.
- Moyes C, Milne A. 1986. Hepatitis B markers in 14–xv year olds in the Bay of Plenty. The New Zealand Medical Journal 99(809): 662–4.
- Rainger W, Solomon Northward, Jones N. 1998. Immunisation coverage and run a risk factors for immunisation failure in Auckland and Northland. New Zealand Public Health Report five(7): 49–51.
- Ramadas D, Moyes CD, Ramadas 1000. 1992. Immunisation status of children in the eastern Bay of Plenty. New Zealand Medical Journal 105(942): 378–9.
- Stehr-Green P, Briasco C, Baker K. 1992. How well are we protecting our children? An immunisation coverage survey in Hawke's Bay. The New Zealand Medical Periodical 105(938): 277–9.
- The Hepatitis Foundation of New Zealand. 2030 Targets. URL: https://www.hepatitisfoundation.org.nz/2030-targets (accessed eleven May 2020).
- Robinson T, Bullen C, Humphries West, et al. 2005. The New Zealand Hepatitis B Screening Plan: screening coverage and prevalence of chronic hepatitis B infection. New Zealand Medical Journal 118(1211): U1345.
- Isle of mann J, Roberts M. 2011. Modelling the epidemiology of hepatitis B in New Zealand. Journal of Theoretical Biology 269(1): 266–72.
- Addidle K. 2011. Bear on of universal hepatitis B vaccination on antenatal hepatitis B prevalence in the Midlands region of the North Island, New Zealand. New Zealand Medical Periodical 124(1332): twoscore–4.
- Lim TH, Gane E, Moyes C, et al. 2015. Serological and clinical outcomes of horizontally transmitted chronic hepatitis B infection in New Zealand Maori: results from a 28-year follow-up study. Gut 64(half dozen): 966–72.
- Lee C, Gong Y, Brok J, et al. 2006. Effect of hepatitis B immunisation in newborn infants of mothers positive for hepatitis B surface antigen: systematic review and meta-assay. BMJ 332(7537): 328–36.
- Van Damme P, Dionne M, Leroux-Roels Chiliad, et al. 2019. Persistence of HBsAg-specific antibodies and immune retention 2 to 3 decades after hepatitis B vaccination in adults. Journal of Viral Hepatitis 26(9): 1066–75.
- European Association for the Report of the Liver. 2017. EASL 2017 Clinical Exercise Guidelines on the management of hepatitis B virus infection. Journal of Hepatology 67(two): p. 370-398.
- Moyes CD, Milne A, Waldon J. 1990. Very low dose hepatitis B vaccination in the newborn: anamnestic response to booster at iv years. Journal of Medical Virology xxx(three): 216–18.
- West DJ, Calandra GB. 1996. Vaccine induced immunologic memory for hepatitis B surface antigen: implications for policy on booster vaccination. Vaccine fourteen(11): 1019–27.
- Van Der Meeren O, Bleckmann G, Crasta PD. 2014. Immune memory to hepatitis B persists in children anile 7–8 years, who were vaccinated in infancy with 4 doses of hexavalent DTPa-HBV-IPV/Hib (Infanrix hexa) vaccine. Human Vaccines & Immunotherapeutics 10(half dozen): 1682–7.
- Su WJ, Liu CC, Liu DP, et al. 2012. Result of age on the incidence of acute hepatitis B subsequently 25 years of a universal newborn hepatitis B immunization program in Taiwan. Journal of Infectious Diseases 205(five): 757–62.
- McMahon BJ, Bulkow LR, Singleton RJ, et al. 2011. Elimination of hepatocellular carcinoma and astute hepatitis B in children 25 years afterward a hepatitis B newborn and catch-up immunization program. Hepatology 54(3): 801–seven.
- Perz JF, Elm JL Jr, Fiore AE, et al. 2006. Virtually elimination of hepatitis B virus infections amidst Hawaii elementary schoolhouse children after universal infant hepatitis B vaccination. Pediatrics 118(4): 1403–8.
- Chen D-S. 2009. Hepatitis B vaccination: The cardinal towards emptying and eradication of hepatitis B. Journal of Hepatology 50(four): 805–xvi.
- Chang M-H. 2011. Hepatitis B virus and cancer prevention. In Senn H-J, Otto F (eds.) Clinical Cancer Prevention. Springer: Berlin & Heidelberg.
- Lee CL, Ko YC. 1997. Hepatitis B vaccination and hepatocellular carcinoma in Taiwan. Pediatrics 99(3): 351–3.
- Whitford K, Liu B, Micallef J, et al. 2018. Long-term impact of baby immunization on hepatitis B prevalence: a systematic review and meta-analysis. Bulletin of the World Health Organization 96(vii): 484–97.
- el-Reshaid Chiliad, al-Mufti S, Johny KV, et al. 1994. Comparison of two immunization schedules with recombinant hepatitis B vaccine and natural amnesty acquired by hepatitis B infection in dialysis patients. Vaccine 12(iii): 223–34.
- Committee on Infectious Diseases. 1994. Update on timing of hepatitis B vaccination for premature infants and for children with lapsed immunisation. Pediatrics 94(three): 403–four.
- American Academy of Pediatrics. 2018. Hepatitis B. In Kimberlin D, Brady M, Jackson M, et al (eds.) Red Book: 2018 Written report of the Committee on Infectious Diseases. URL: https://redbook.solutions.aap.org/redbook.aspx (accessed three July 2020).
- Sarin SK, Kumar M, Lau GK, et al. 2016. Asian-Pacific clinical practise guidelines on the management of hepatitis B: a 2015 update. Hepatology International 10(one): p. ane–98.
- Terrault NA, Bzowej NH, Chang KM, et al. 2016. AASLD guidelines for treatment of chronic hepatitis B. Hepatology 63(1): p. 261–83.
- Roukens AH, Visser LG. 2011. Hepatitis B vaccination strategy in vaccine low and non-responders: a matter of quantity of quality? Hum Vaccin seven(6): 654–vii.
- Hepatitis Foundation of New Zealand. 20202. Hepatitis B for Health Professionals. URL: https://www.hepatitisfoundation.org.nz/health-professionals/hepatitis-b-health-professionals (accessed 20 January 2020).
- European Consensus Group on Hepatitis B Amnesty. 2000. Are booster immunisations needed for lifelong hepatitis B immunity? European Consensus Group on Hepatitis B Immunity. Lancet 355(9203): 561–5.
- Raven SFH, Hoebe C, Vossen A, et al. 2020. Serological response to three alternative series of hepatitis B revaccination (Fendrix, Twinrix, and HBVaxPro-twoscore) in healthy non-responders: a multicentre, open-characterization, randomised, controlled, superiority trial. Lancet Infectious Diseases twenty(1): 92–101.
- Cardell K, Akerlind B, Sallberg M, et al. 2008. Splendid response rate to a double dose of the combined hepatitis A and B vaccine in previous nonresponders to hepatitis B vaccine. Journal of Infectious Diseases 198(3): 299–304.
- Bohlke Thou, Davis RL, Marcy SM, et al. 2003. Risk of anaphylaxis after vaccination of children and adolescents. Pediatrics 112(4): 815–20.
- Establish of Medicine: Commission to Review Adverse Furnishings of Vaccines. 2012. Adverse Effects of Vaccines: Evidence and causality (ed). Washington, DC: The National Academies Printing. URL: https://www.nap.edu/catalog/13164/adverse-furnishings-of-vaccines-evidence-and-causality (accessed January 2020).
- Expanded Programme on Immunization (EPI). 1997. Expanded programme on immunization (EPI): Lack of testify that hepatitis B vaccine causes multiple sclerosis. Weekly Epidemiological Tape 72(21): 149–52.
- Mouchet J, Begaud B. 2019. Hepatitis B vaccination and central demyelination: history, clarification and observed/expected analyses of 624 cases reported to the French pharmacovigilance over a xx-year period. Vaccine 37(xv): 2142–8.
- Sadovnick Advertising, Scheifele DW. 2000. School-based hepatitis B vaccination plan and adolescent multiple sclerosis. Lancet 355(9203): 549–50.
- Health and Welfare Canada. 1993. Report of the working group on the possible relationship between hepatitis B vaccination and the chronic fatigue syndrome. Canadian Communicable Disease Report xix(4): 25–8.
- Wise RP, Kiminyo KP, Salive ME. 1997. Pilus loss after routine immunizations. JAMA 278(14): 1176–8.
- Ronchi F, Cecchi P, Falcioni F, et al. 1998. Thrombocytopenic purpura equally adverse reaction to recombinant hepatitis B vaccine. Archives of Disease in Childhood 78(iii): 273–4.
- Fisher MA, Eklund SA, James SA, et al. 2001. Agin events associated with hepatitis B vaccine in US children less than half dozen years of age, 1993 and 1994. Annals of Epidemiology 11(1): thirteen–21.
- McMahon BJ, Helminiak C, Wainwright RB, et al. 1992. Frequency of adverse reactions to hepatitis B vaccine in 43,618 persons. American Journal of Medicine 92(3): 254–half dozen.
- Ni YH, Chang MH, Wu JF, et al. 2012. Minimization of hepatitis B infection by a 25-year universal vaccination program. Journal of Hepatology 57(four): 730–5.
- Romanò L, Paladini S, Van Damme P, et al. 2011. The worldwide impact of vaccination on the control and protection of viral hepatitis B. Digestive and Liver Disease 43(Suppl 1): S2–vii.
- Ministry building of Wellness. 2012. Hepatitis B. in Communicable Disease Control Manual. Wellington. URL: http://www.health.govt.nz/publication/communicable-disease-command-transmission-2012 (accessed 25 Feb 2020).
williamstores1988.blogspot.com
Source: https://www.health.govt.nz/our-work/immunisation-handbook-2020/9-hepatitis-b
0 Response to "How Long Have Babies Been Getting the Hepatitis B Vaccine"
Post a Comment